Temperature- the quantity that tells how hot or cold something is compared with a standard.
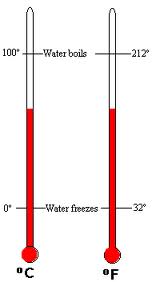
Celsius scale- 0 to 100 degrees
Fahrenheit scale- 32-312 degrees scale
Fahrenheit scale- 32-312 degrees scale
Kelvin scale- its degrees are the same size as the Celsius degree and are called "kelvins"
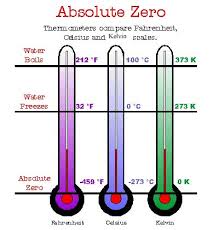
Absolute zero- a substance that has no kinetic energy to give up

Heat- the energy thats transfers from one object to another bacause of a temperature difference between them
Thermal Contact- when heat flows from one object or substance to another it is in contact with
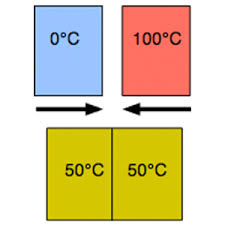
Thermal Equilibrium-after objects in thermal contact with each other reach the same temperature, no heats flow between them

_internal energy- the grand total of all energies inside a substance

Calorie- the amount of heat required to raise the temperature of 1 gram of by 1C

Kilocalorie- 1000 calories

Specific heat capacity- when water absorbs more heat per gram than iron for the same change in temperature.

Bimetallic strip- two stripes of different metals are welded or riveted together.

Thermostat- a practical application of a bimetallic strip

Conduction

Convection

Radiation

Joules

Calories

1st Law of Thermodynamics
